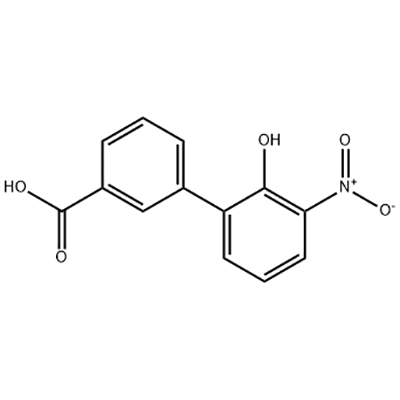
Newly Arrival Reverse Osmosis Filter System - 2′-hydroxy-3′-nitro-3-biphenylcarboxylic acid – JIN DUN
Newly Arrival Reverse Osmosis Filter System - 2′-hydroxy-3′-nitro-3-biphenylcarboxylic acid – JIN DUN
Newly Arrival Reverse Osmosis Filter System - 2′-hydroxy-3′-nitro-3-biphenylcarboxylic acid – JIN DUN Detail:
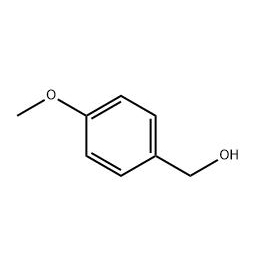
2′-hydroxy-3′-nitro-3-biphenylcarboxylic acid is used as the intermediate of Eltrombopag .
Eltrombopag, developed by GlaxoSmithKline (GSK) in the UK and later jointly developed with Novartis in Switzerland, is the first and only approved small molecule non peptide TPO receptor agonist in the world. Eltrombopag was approved by the US FDA in 2008 for the treatment of idiopathic thrombocytopenic purpura (ITP), and in 2014 for the treatment of severe aplastic anemia (AA). It is also the first drug approved by the US FDA for the treatment of AA in recent 30 years.
In december2012, the US FDA approved Eltrombopag for the treatment of thrombocytopenia in patients with chronic hepatitis C (CHC), so that hepatitis C patients with poor prognosis due to low platelet count can start and maintain interferon based standard therapy for liver diseases. On february3,2014, GlaxoSmithKline announced that the FDA granted the breakthrough treatment drug qualification of Eltrombopag for the treatment of hemopenia in patients with severe chemicalbook aplastic anemia (SAA) who did not fully respond to immunotherapy. On August 24, 2015, the US FDA approved Eltrombopag for the treatment of thrombocytopenia in adults and children aged 1 year and over with chronic immune thrombocytopenia (ITP) who have insufficient response to corticosteroids, immunoglobulins or splenectomy. On january4,2018, Eltrombopag was approved to be listed in China for the treatment of primary immune thrombocytopenia (ITP).
Product detail pictures:
Related Product Guide:
With our excellent management, strong technical capability and strict quality control system, we continue to provide our clients with reliable quality, reasonable prices and excellent services. We aim at becoming one of your most reliable partners and earning your satisfaction for Newly Arrival Reverse Osmosis Filter System - 2′-hydroxy-3′-nitro-3-biphenylcarboxylic acid – JIN DUN , The product will supply to all over the world, such as: Turkey, Toronto, Thailand, With the spirit of "credit first, development through innovation, sincere cooperation and joint growth", our company is striving to create a brilliant future with you, so as to become a most valuable platform for exporting our goods in China!
Good quality, reasonable prices, rich variety and perfect after-sales service, it's nice!



![factory low price Inhibitor Mehq - 6-tetra-O-acteyl-1-C-[4-chloro-3-[[4-[[(3S)-tetrahydrofu-ran-3-yl]oxy]phenyl] – JIN DUN](https://cdn.globalso.com/jindunchem-med/0ecf55f0.jpg)