2-butyl-5-nitro-3-benzofuranyl)[4-[3-(dibutylaMino)propoxy]phenyl]
2-butyl-5-nitro-3-benzofuranyl)[4-[3-(dibutylaMino)propoxy]phenyl]
It is clinically used in primary and secondary Hyperuricemia, especially in patients with hyperuricemia, as well as in patients with renal insufficiency, and in patients with recurrent or chronic gout, as well as in patients with Hyperuricemia. Used in patients with Gouty nephropathy to relieve symptoms and reduce the formation of renal uric acid stones; gout stones; used in uric acid kidney stones and uric acid kidney disease.
The new anti-arrhythmic drug Atrial fibrillation (AF) is a potentially life-threatening disease. Due to the aging of the population, the global incidence and mortality of atrial fibrillation are increasing, and it has become a new public Health problems. There are 2.5 million patients with atrial fibrillation in the United States and 4.5 million in the European Union. According to statistics, the prevalence of atrial fibrillation among people over 30 years old in my country is 0.77%, which increases with age, and males are higher than females (0.9%:0.7%). The diseases that cause atrial fibrillation mainly include hypertension, coronary heart disease, valvular heart disease, spontaneous dilated cardiomyopathy, and heart failure. The main symptom of patients with atrial fibrillation is irregular ventricular rate, and heart rate control occupies a certain position in the acute treatment and maintenance of symptoms. Amiodarone plays a good role in controlling heart rate and restoring sinus rhythm, but the adverse effects of target organs related to the iodine component in its structure limit its clinical application, such as damage to the lung, thyroid, and liver. The above-mentioned adverse reactions and its structure The iodine in is closely related. The new antiarrhythmic drug Dronedarone (Dronedarone) was obtained by modifying the structure of amiodarone. Dronedarone has electrophysiological properties similar to amiodarone, but does not contain iodine in its structure, so it can avoid serious extra-cardiac side effects caused by iodine Chemicalbook. Dronedarone was approved for marketing by the U.S. Food and Drug Administration (FDA) in July 2009, and was approved for marketing in the European Union in 2009. It is also the first new antiarrhythmic drug approved in the European Union in 10 years. Dronedarone is N-[2-butyl-3-[4-[3-(dibutylamino)propoxy]phenyl]-5-benzofuranyl]-methanesulfonamide, which is to reduce A benzofuran derivative obtained by structural modification of amiodarone, which is not a cardiac adverse reaction. It removes the iodine atom in amiodarone to reduce the toxicity of the thyroid and other organs; adds a methylsulfonyl group on the benzofuran side to reduce lipophilicity, thereby shortening the half-life of the drug and reducing the tissue accumulation effect and possibility of the drug Of neurotoxicity. Both the 2011 US version and the 2010 European version of the Atrial Fibrillation Guidelines recommend dronedarone for the treatment of atrial fibrillation. Dronedarone is safe and effective in treating ventricular rate and maintaining sinus rate in various patients without severe AF. It can be an effective drug to replace amiodarone. Dronedarone can also reduce the death of AF patients and even persistent AF patients. Therefore, it is expected to become the first-line medication for AF patients without severe heart failure. Moreover, the use of anticoagulants in middle-high-risk AF patients is beneficial to the use of dronedarone.
Use:Intermediate for Dronedarone hydrochloride
Standard:enterprise standard
Assay:≥98%
Exterior: Yellow oily liquid
Package: 25kg/drum

JIN DUN Medical has ISO qualification and meets GMP production standards, employed domestic and foreign drug synthesis experts with rich experience to guide company's R&D.




TECHNOL OGY ADVANTAGES
●High Pressure Catalytic Hydrogenation . High Pressure Hydrogenolysis Reaction . Cryogenic Reaction (<-78%C)
●Aromatic Heterocyclic Synthesis
●Rearrangement Reaction
●Chiral Resolution
●Heck, Suzuki,Negishi,Sonogashira . Gignard Reaction
Equipments
Our Lab has various experimental and testing equipments, such as: NMR (Bruker 400M)、HPLC、chiral-HPLC、LC-MS、LC-MS/MS (API 4000)、IR、UV、GC、GC-MS, Chromatography, Microwave Synthesizer, Parallel Synthesizer, Differential Scanning Calorimeter (DSC), Electron Microscope...
R&D Team
Jindun Medical has a group of professional R&D personnel, and employs many domestic and foreign drug synthesis experts to guide R&D, making our synthesis more accurate and efficient.

We have helped several top domestic pharmaceutical companies, such as Hansoh, Hengrui and HEC Pharm. Here we will show part of them.
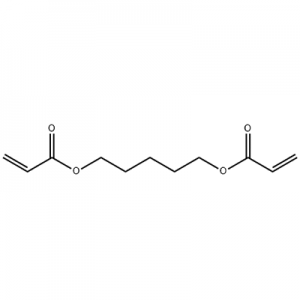
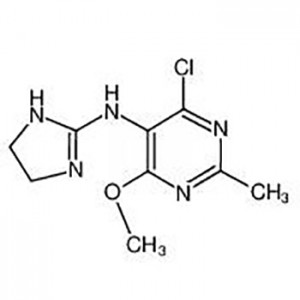
Customzation Case One:
Cas No.: 110351-94-5
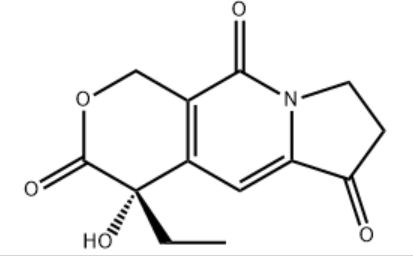
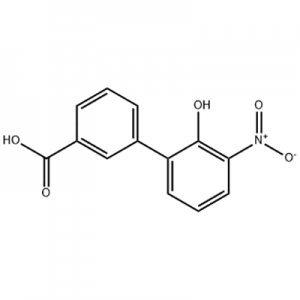
Customzation Case Two:
Cas No.: 144848-24-8
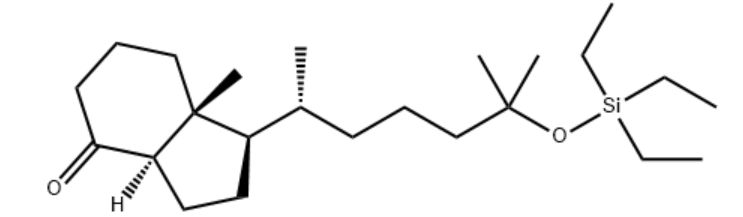
Customzation Case Three:
Cas No.: 200636-54-0
1.Customize New Intermediates or APIs. Same as above case sharing, customers have the demands for specific Intermediates or APIs, and they can not find the needed products in the market, then we can help to Customize.
2.Process Optimization for Old Products. Our team will help to Optimize and improve such production whose reaction route is old, production cost is high, and the efficiency is low. We can provide full documentation for technology transfer and process improvement, help customer for a more efficient production.
From drug targets to INDs, JIN DUN Medical provides you with one-stop personalized R&D solutions.
JIN DUN Medical insists on creating a team with dreams, making dignified products, meticulous, rigorous, and going all out to be a trusted partner and friend of customers!


![2-butyl-5-nitro-3-benzofuranyl)[4-[3-(dibutylaMino)propoxy]phenyl] Featured Image](https://cdn.globalso.com/jindunchem-med/922e79ba.jpg)

![5-[2-cyclopropyl-1-(2-fluorophenyl)-2-oxoethyl]- 5,6,7,7a-tetrahydrothieno[3,2-c]pyridin-2 (4h)-one](https://cdn.globalso.com/jindunchem-med/image41-300x300.png)